By 2009, only 26 manufacturers worldwide produced carbon tetrachloride, including three in the United States. production and export of CCl 4 dropped precipitously during this time. By 2009, the United States no longer regularly imported CCl 4 only 90 pounds (41 kilograms) were imported during 1996-2009. īoth the internationally ratified Montreal Protocol (which first went into effect in 1989) and the United States 1990 Clean Air Act Amendments were instrumental in reducing environmental concentrations of CCl 4 and other ozone-depleting chemicals. It is not permitted in products intended for home use however, chemicals containing CCl 4 can still be purchased online. In the United States, CCl 4 use is restricted to industrial and laboratory applications only. a) Na, F b) Be, O C) Al, S d) K, Se e) Li, N f) Mg, P g) Ca, H Give the correct chemical formula for each of the following binary compounds: nonmetal-nonmetal. Environmental Protection Agency cancelled CCl 4‘s use as a fumigant in 1986. Write the chemical formula(s) of the ionic compounds formed between each of the following pairs (metal and nonmetal) of elements. CCl 4 was also used as an antihelminthic, a grain fumigant, and a component in fire extinguishers.Ĭarbon tetrachloride’s toxicity was recognized in the early 1900s, and most of the uses were discontinued by the mid-1960s. It was found in household cleaning supplies and spot removers for carpets, clothing, and furniture. In the United States, CCl 4 was also an ingredient in many industrial fluids, and was an effective metal degreaser. It can also cause damage to eyes and it is harmful to animals and environments, it cause damage to the ozone layer as well.Historically, CCl 4 was mainly used to produce chlorofluorocarbons (CFCs), which are used as heat transfer agents in refrigerating equipment and as aerosol propellants. Carbon tetrachloride is suspected to be a human carcinogen. It can cause degeneration of liver and kidneys. Health effects / safety hazards: Carbon tetrachloride is toxic and poisoning by inhalation or ingestion. Carbon tetrachloride is also used as cleaning agent and refrigerant in low concentrations. This compound is often classified as a polyhalogenated organic compound since it consists of a carbon atom which is attached to more than one halide functional group. It is also a solvent for some reactions in laboratory scale, however, its application is reduced due to it is poisoning by inhalation. What is Carbon Tetrachloride Carbon tetrachloride, also known as tetrachloromethane, is an organic compound with the chemical formula CCl4. Uses: Carbon tetrachloride is used to produce a wide variety of chemical compounds such as oils, lacquers, rubbers, resins and coats. 
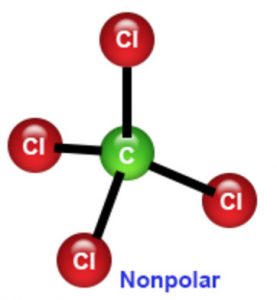
Consequently, carbon tetrachloride is a solvent that can only be used to dissolve non-polar or low polarity compounds, such as fats and oils, thus it is used in the cleaning agents manufacturing. These bonds have dipolar vectors that are cancelled, resulting in a polarity of zero. It is not soluble in water, but it is soluble in ethanol, methanol, benzene, ether and hexane.Ĭhemical properties: Carbon tetrachloride is a non-polar molecule because its symmetrical structure formed by 4 C-Cl bonds. Carbon tetrachloride melting and boiling points are -22.92 ✬ and 76, 7 ✬, respectively. Physical properties: Carbon tetrachloride is a colorless liquid with a sweet, ether-like odor, Its density is 1.5867 g mL-1. 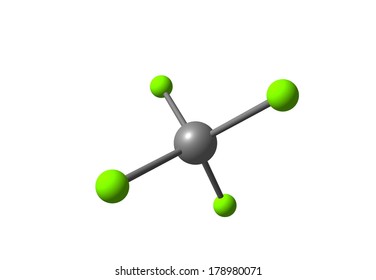
Using the reaction of sulfur monochloride with cabon disulfide. Through the Lummus oxychlorination process with natural gas and HCl in its gas form The reaction of pyrolysis of hexachloroethane. By the reaction of chlorination of methane, which also produces other chlorides as CHCl 3, CH 2Cl 2 and CH 3Cl
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
